 A number of neutrons are 14 and it belongs to the third period now. Atomic number is 13, has 13 plotons, 13 electrons, it has atomic mass of 26.982 and it has around atomic mass 27. The next element is the alminium alminium. We have to subtract mass number from protons, so it comes to be 6 and it belongs to the second period. Rounded off mass number is 12 point, so its number of neutrons are. It has 6 protons 6 electrons, as number is 12.0 11. Is the son has a symbol c: its atomic number is 6. The number of protons, so it comes to be 2, which belongs to the first period now the next given element is the next given element. The first element is the helium first element is the helium, so the simple used for helium is its atomic number, is 2 number of protons are 2? Electrons are 2? Mass number is 4.0 2 rounded, as 4 number of neutrons are a number of mass number minus. Then their atomic number than the number of protons present in it than the number of electrons present in it than the atomic mass number than the rounded rounded mass number than the neutrons present in it. Holden 1985 3 p refs Presented at the 33rd IUPAC General. The elements that are given in the question that must be written here is the symbols that used to designate them. However, the rounding up rule will be discontinued and the. Number of protons so the elements so the given elements are firstly here. There are certain elements that are given in the question and we have to find their symbols. 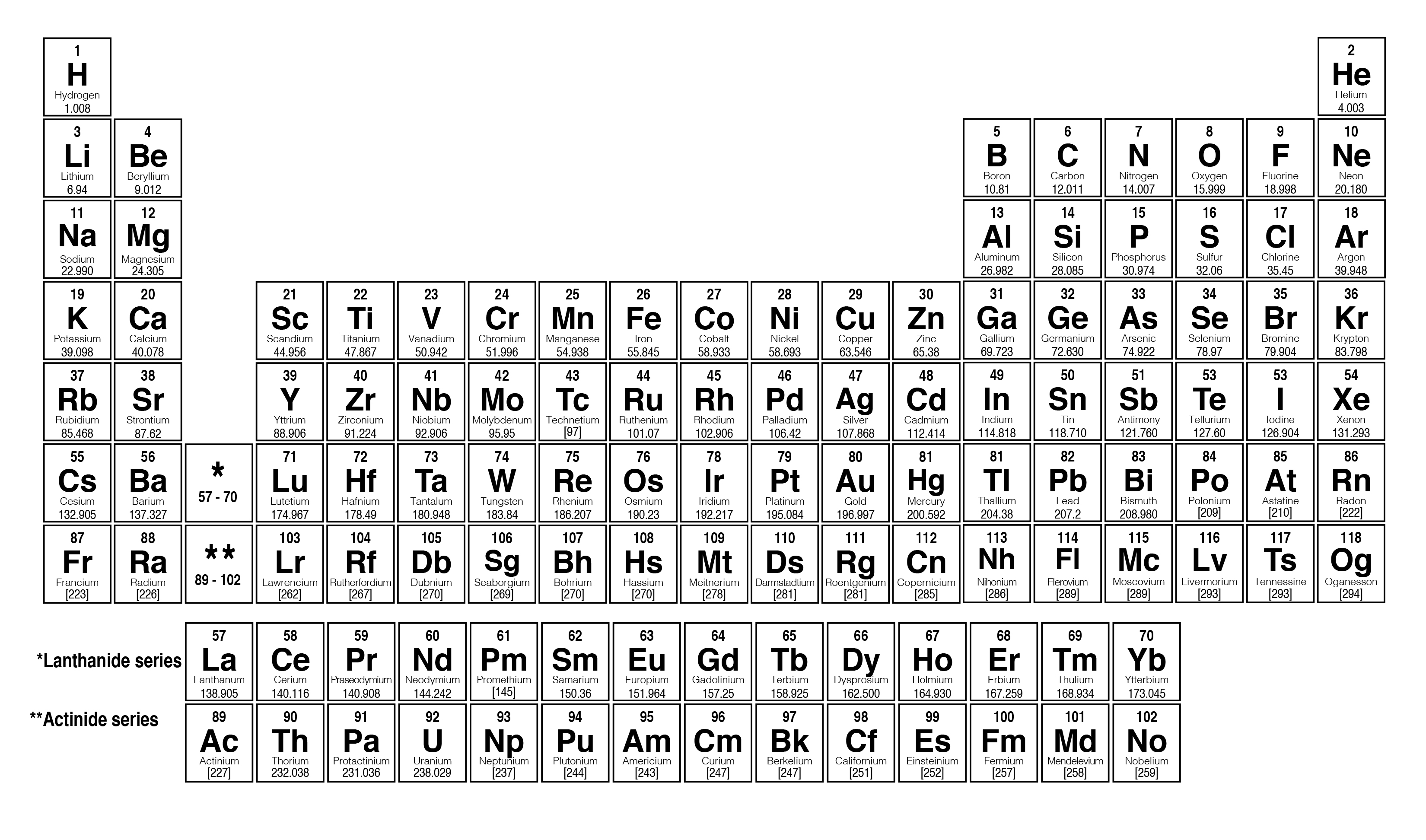
The elements are arranged according to their groups and periods according to their chemical properties. The details all of the elements in the Periodic table, the numbers of protons, the numbers of neutrons and the mass numbers of atoms which relate to the elements in the Periodic Table.Hello students- let's solve this question, this question is based to the periodic table in periodic table. The mass number (represented by the letter A) is defined as the total number of protons and neutrons in an atom. Neon has the Atomic Mass weight of 20.18. The number of protons in atom of neon is therefore 10. Example 3 - mass number of Neon: The element Neon (Symbol Ne) has the Atomic Number of 10.The mass number of silver is therefore 108. Silver has the Atomic Mass weight of 107.87. The number of protons in atom of silver is therefore 47. (262) Interactive periodic table of elements - your complete guide to the elements including definition, mass & names of each chemical in the periodic table. Example 2 - mass number of Silver: The element Silver (Symbol Ag) has the Atomic Number of 47.The mass number of gold is therefore 197. Gold has the Atomic Mass weight of 196.97. The number of protons in atom of gold is therefore 79. Example 1 - mass number of Gold: The element Gold (Symbol Au) has the Atomic Number of 79.The following examples provide details of how to calculate the mass number. The unique chart below has been created by and details all of the elements in the Periodic table, the numbers of protons, the numbers of neutrons and the mass numbers of atoms which relate to the elements. So, if we know the number of protons and neutrons in an atom we can determine the mass number. Mass Numbers - The Mass Numbers of all of the elements The atomic mass is carried by the atomic nucleus, which occupies only about 10 -12 of the total. :max_bytes(150000):strip_icc()/PeriodicTableSigFigBW-58b5c7f25f9b586046cae098.png)
Number of Neutrons = Mass Number - Atomic Number Mass Numbers The atomic mass or relative isotopic mass refers to the mass of a single particle, and therefore is tied to a certain specific isotope of an element. Mass Numbers = Atomic Weight of Element, rounded to nearest whole number Subtracting the Atomic number from the Mass Number equals the number of neutrons in the nucleus. Mass numbers equal the total number of heavy, or massive, particles in the nucleus. /PeriodicTable-58b5d9253df78cdcd8d03ebb.jpg) 
The chemical properties of an element are determined by its Atomic Number not its Mass Number which is why atomic numbers are shown on the Periodic table whilst Mass Numbers are not. The Periodic Table with Atomic Mass will give you the atomic weight, or atomic mass, of the elements. The mass number is established by rounding the atomic weight to the nearest whole number. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
